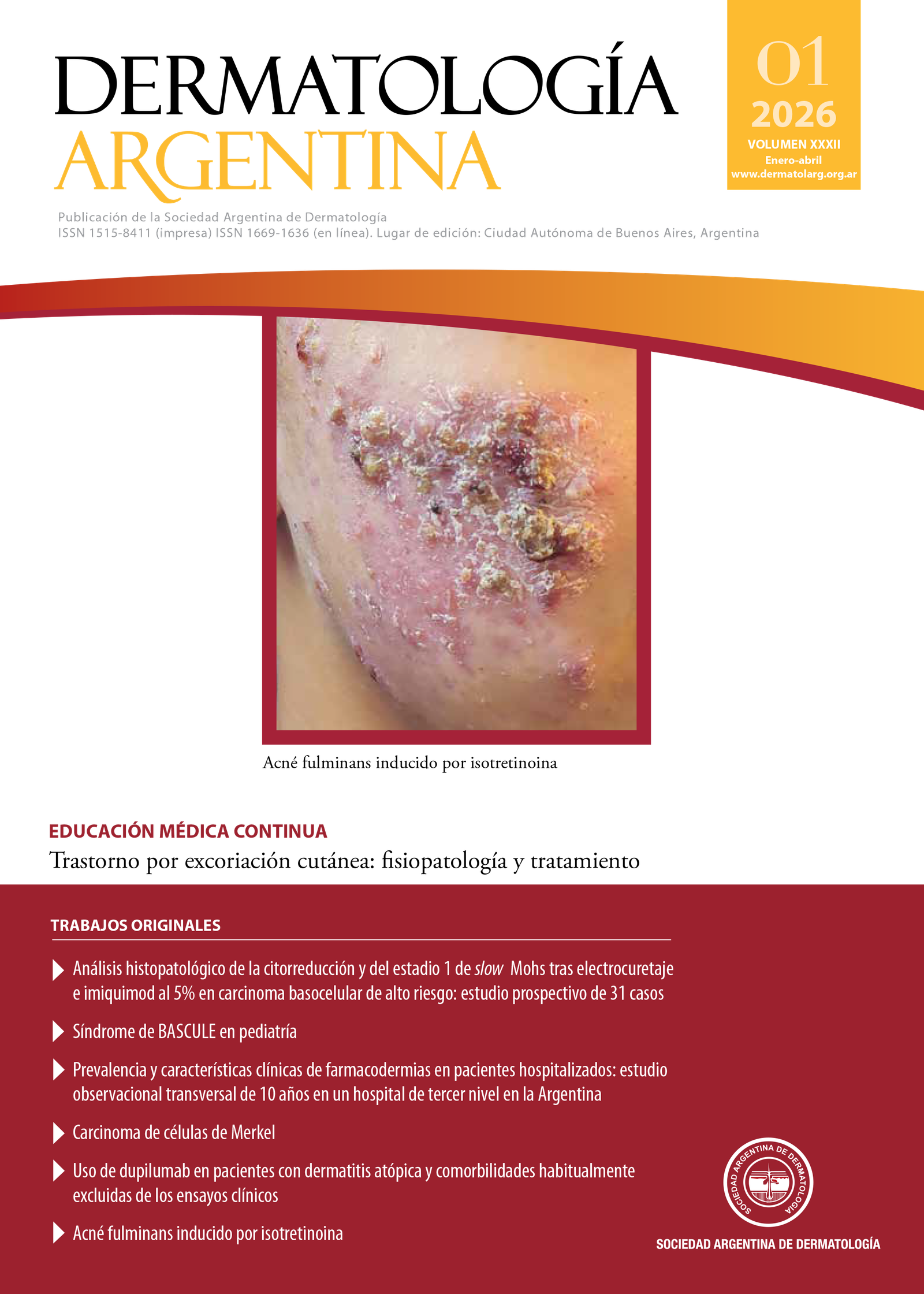
Histopathological analysis of cytoreduction and slow Mohs stage 1 after electrocurettage and 5% imiquimod in high-risk basal cell carcinoma: a prospective study of 31 cases
DOI:
https://doi.org/10.47196/3zppmk55Keywords:
curettage, imiquimod, Slow Mohs, basal cel carcinoma, high-risk, cytoreductionAbstract
Introduction: curettage (EC) and 5% imiquimod are effective therapies for low-risk basal cell carcinoma (BCC). In high-risk cases, prior use of imiquimod has been shown to reduce the number of Mohs stages. There are no studies evaluating the combination of EC + 5% imiquimod with histopathological analysis of cytoreduction and the first stage of Slow Mohs.
Objectives: To analyze the histopathology of cytoreduction and the first stage of Slow Mohs after EC + 5% imiquimod in high-risk BCC and to compare the number of stages with a historical institutional series without pretreatment.
Materials y methods: a prospective, quasi-experimental study. Thirty-one high-risk BCCs treated between August 2024 and March 2025 were included. Patients received EC followed by 5% imiquimod for six weeks and subsequently underwent Slow Mohs. Tumor persistence was defined as positive cytoreduction or first stage.
Results: tumor absence was observed in 87.1% (27/31; 95% CI: 69-96%) while tumor persistence was 12.9% (4/31; 95% CI: 4.2-31%). In the nose (n=15) persistence was 26.7%, whereas in extranasal locations (n=16) it was 0%.
Conclusions: the combination of curettage + 5% imiquimod before slow Mohs surgery showed a high rate of histopathological negativity, especially outside the nose, suggesting potential savings in stages and usefulness in contexts without access to Mohs surgery or with contraindications.
References
I. Schmults CD, Blitzblau R, Aasi SZ, Alam M, et al. Basal cell skin cancer, Version 2.2024, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2023;21:1181-1203.
II. Peris K, Fargnoli MC, Garbe C, Kaufman R, et al. Diagnosis and treatment of basal cell carcinoma: European consensus-based interdisciplinary guidelines. Eur J Cancer. 2019;118:10-34.
III. Kim JYS, Kozlow JH, Mittal B, Moyer J, Olencki T, Rodgers P, et al.; Work Group; Invited reviewers. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018 Mar;78(3):540-59.
IV. Chanal J. Prise en charge du carcinome basocellulaire en 2025 [Management of basal cell carcinoma in 2025]. Ann Chir Plast Esthet. 2025;70:452-456.
V. Marzuka AG, Book SE. Basal cell carcinoma: pathogenesis, epidemiology, clinical features, diagnosis, histopathology, and management. Yale J Biol Med. 2015;88:167-179.
VI. Cameron MC, Lee E, Hibler BP, Barker C, et al. Basal cell carcinoma. Epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317.
VII. Filho LL, de Oliveira de Avelar Alchorne A, Pereira GC, Lopes LR, et al. Histological and immunohistochemical evaluation of basal cell carcinoma following curettage and electrodessication. Int J Dermatol. 2008;47:610-614.
VIII. Ezughah FI, Affleck AG, Evans A, Ibbotson SH, et al. Confirmation of histological clearance of superficial basal cell carcinoma with multiple serial sectioning and Mohs' micrographic surgery following treatment with imiquimod 5% cream. J Dermatolog Treat. 2008;19:156-158.
IX. Aghajani M, Savage A, Bonilla GM. Imiquimod 5% cream as an adjunct to Mohs micrographic surgery in basal cell carcinoma. A mixed methods systematic review. Dermatol Surg. 2025;51:847-851.
X. Sutrisno CSN, Pramita DH, Dewi IP. Curative or conservative approaches. A systematic review of surgical and nonsurgical treatments for basal cell carcinoma. Cureus. 2025;17:e95556.
XI. van der Geer S, Martens J, van Roij J, Brand E, et al. Imiquimod 5% cream as pretreatment of Mohs micrographic surgery for nodular basal cell carcinoma in the face: a prospective randomized controlled study. Br J Dermatol. 2012;167:110-115.
XII. Williams HC, Bath-Hextall F, Ozolins M, Amstrong S, et al. Surgery versus 5% imiquimod for nodular and superficial basal cell carcinoma: 5-year results of the SINS randomized controlled trial. J Invest Dermatol. 2017;137:614-619.
XIII. Verkouteren BJA, Nelemans PJ, Sinx KAE, Kelleners-Smeets N, et al. Imiquimod cream preceded by superficial curettage versus surgical excision for nodular basal cell carcinoma: a secondary analysis of a randomized clinical trial. JAMA Dermatol. 2025;161:299-304.
XIV. Lawrence CM, Haniffa M, Dahl MG. Formalin-fixed tissue Mohs surgery (slow Mohs) for basal cell carcinoma: 5-year follow-up data. Br J Dermatol. 2009; 160:573-580.
XV. Asilian A, Moeine R, Hafezi H, Shahriarirad R. Treatment of vulvar basal cell carcinoma with Slow-Mohs micrographic surgery. A case report. Clin Case Rep. 2022;10:e6442.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 On behalf of the authors. Reproduction rights: Argentine Dermatology

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
El/los autor/es tranfieren todos los derechos de autor del manuscrito arriba mencionado a Dermatología Argentina en el caso de que el trabajo sea publicado. El/los autor/es declaran que el artículo es original, que no infringe ningún derecho de propiedad intelectual u otros derechos de terceros, que no se encuentra bajo consideración de otra revista y que no ha sido previamente publicado.
Le solicitamos haga click aquí para imprimir, firmar y enviar por correo postal la transferencia de los derechos de autor